Product Description
| Download Documents | |
| SDS | USER MANUAL will be supplied to customer |
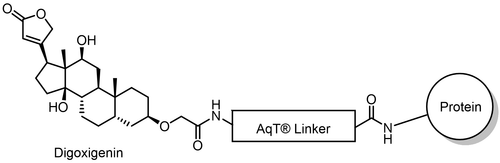
AqT® Digoxigenin Antibody Labeling kit is a proprietary digoxigenin labeling kit developed at CellMosaic® for obtaining high quality and high loading of digoxigenin labeled antibody with minimal or no aggregation.
For a detailed description of the AqT® technologies, please see our technology section.
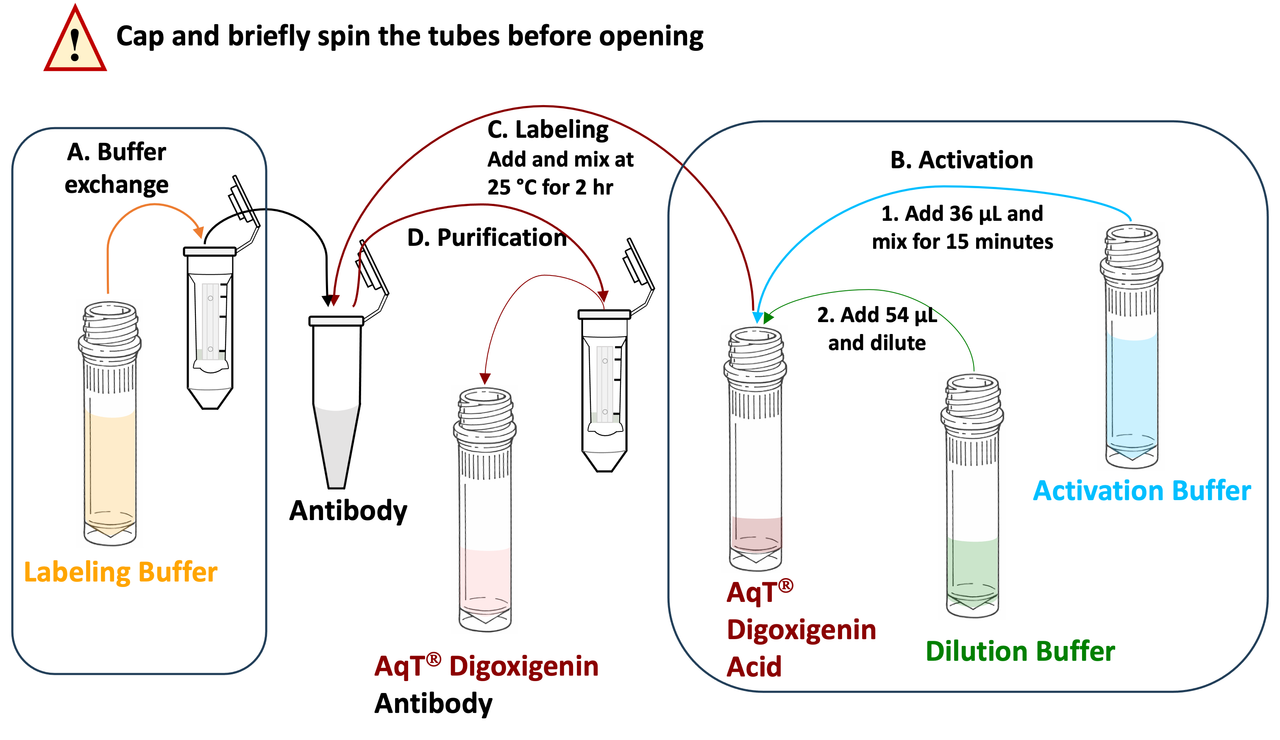
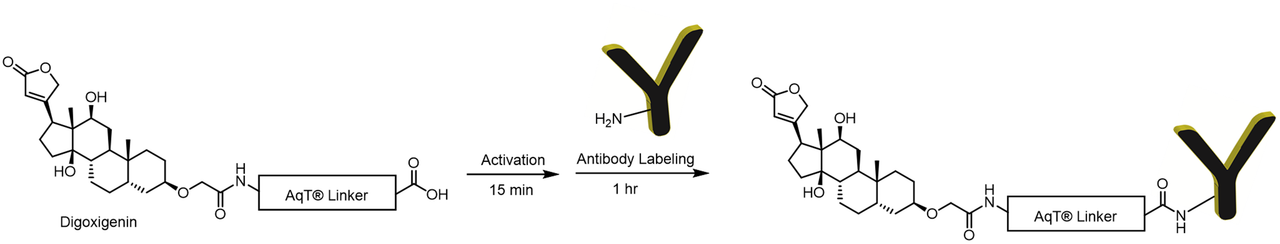
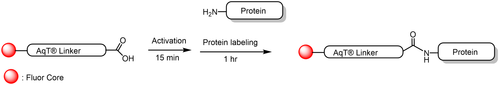
The CM86440 kit is designed to label any antibody with AqT® digoxigenin via surface amines on the antibody. The user supplies the antibody. The kit includes AqT® digoxigenin acid, which is activated within 15 minutes and then coupled directly to the antibody in one step within 1 hour. The labeled product is subsequently purified to remove any unreacted AqT® digoxigenin acid
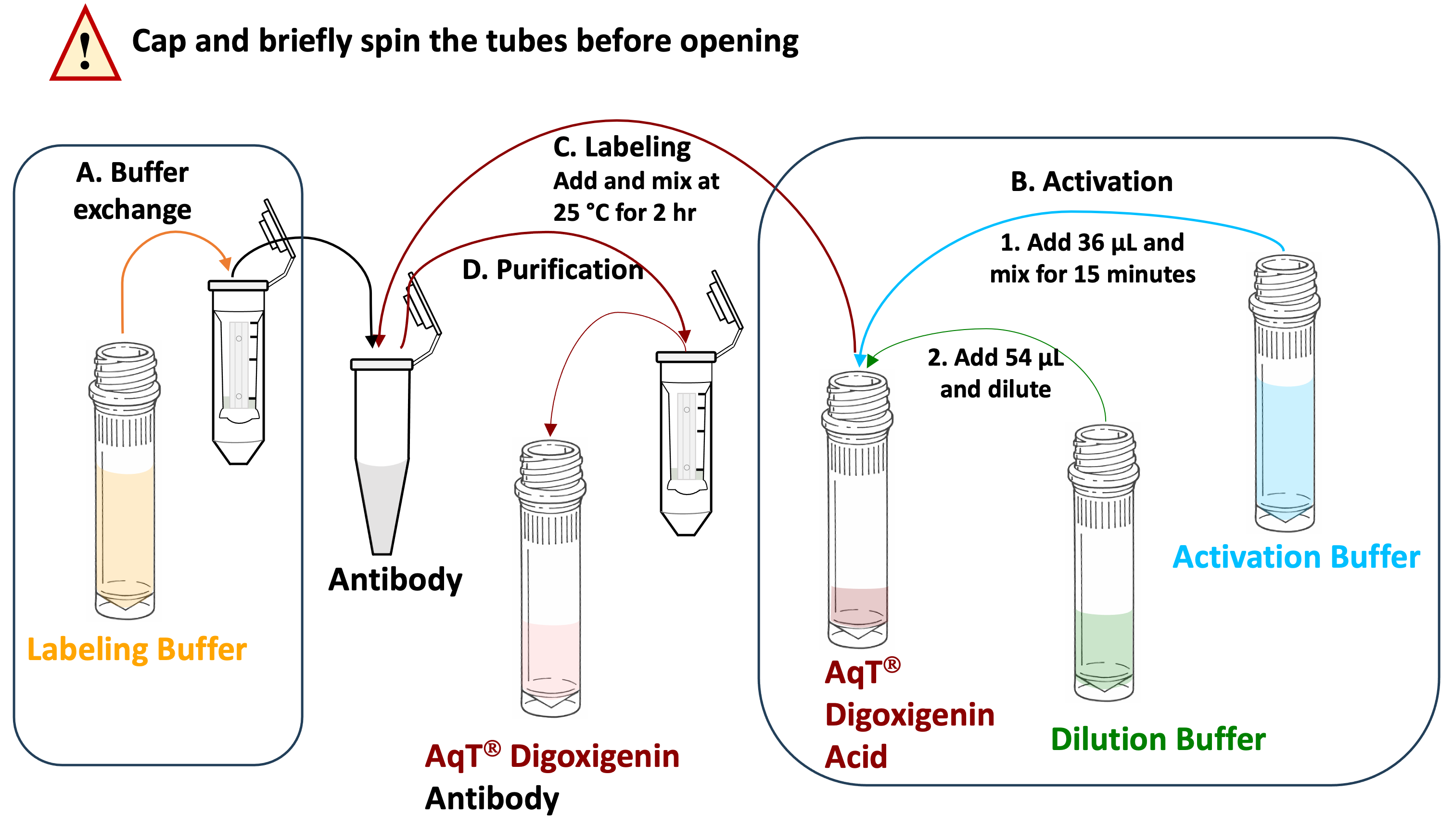
The kit is available in multiple configurations for labeling antibodies ranging from 0.1 to 3 mg. Each configuration supports labeling of either one (×1) or three (×3) antibody samples. Please refer individual configuration to use the user manual.
|
Catalog Number |
Number of Reactions in Kit |
Antibody Amount per Reaction |
| CM86440.01x1 | 1 | 100 μg (0.667 nmol for IgG) |
| CM86440.01x3 | 3 | |
| CM86440.1x1 | 1 | 0.1-1 mg (0.667-6.67 nmol for IgG) |
| CM86440.1x3 | 3 | |
| CM86440x1 | 1 | 1-3 mg (6.67-20 nmol for IgG) |
| CM86440x3 | 3 |
If you have any other customization needs, please provide the details above.
Requirement for Antibody
- MW: ≥ 100 KDa (if MW is lower, please use an alternative kit)
- Preferably >90% pure by gel electrophoresis
- Total amount: 0.1-3 mg of protein content as measured by UV.
Key Features
- Advanced AqT® linker technology: Utilizes CellMosaic’s proprietary, super-hydrophilic, water-soluble, charge-neutral AqT® linker to minimize digoxigenin stacking and antibody aggregation.
- High digoxigenin loading with preserved antibody properties: Typical degree of labeling (DOL) of 6–8 digoxigenin per IgG antibody for increased sensitivity, with minimum aggregation..
- All-inclusive kit with validated analytical method: Provides sterilized buffers and tubes, and filters for digoxigenin labeling. Also includes method needed for concentration measurement and loading determination.
- Fast, user-friendly workflow plus optional support: One-step labeling and purification in ~2 hours (≤1 hour hands-on), with optional post-conjugation services at CellMosaic® for analytical characterization and DOL determination.
Labeling Chemistry

Protocol
Schematic workflow diagram for preparing AqT® digoxigenin labeled antibody within 2 hours, starting with 3 mg of antibody. (Reagent volume will vary if the amount of IgG is less than 3 mg).
Example Data
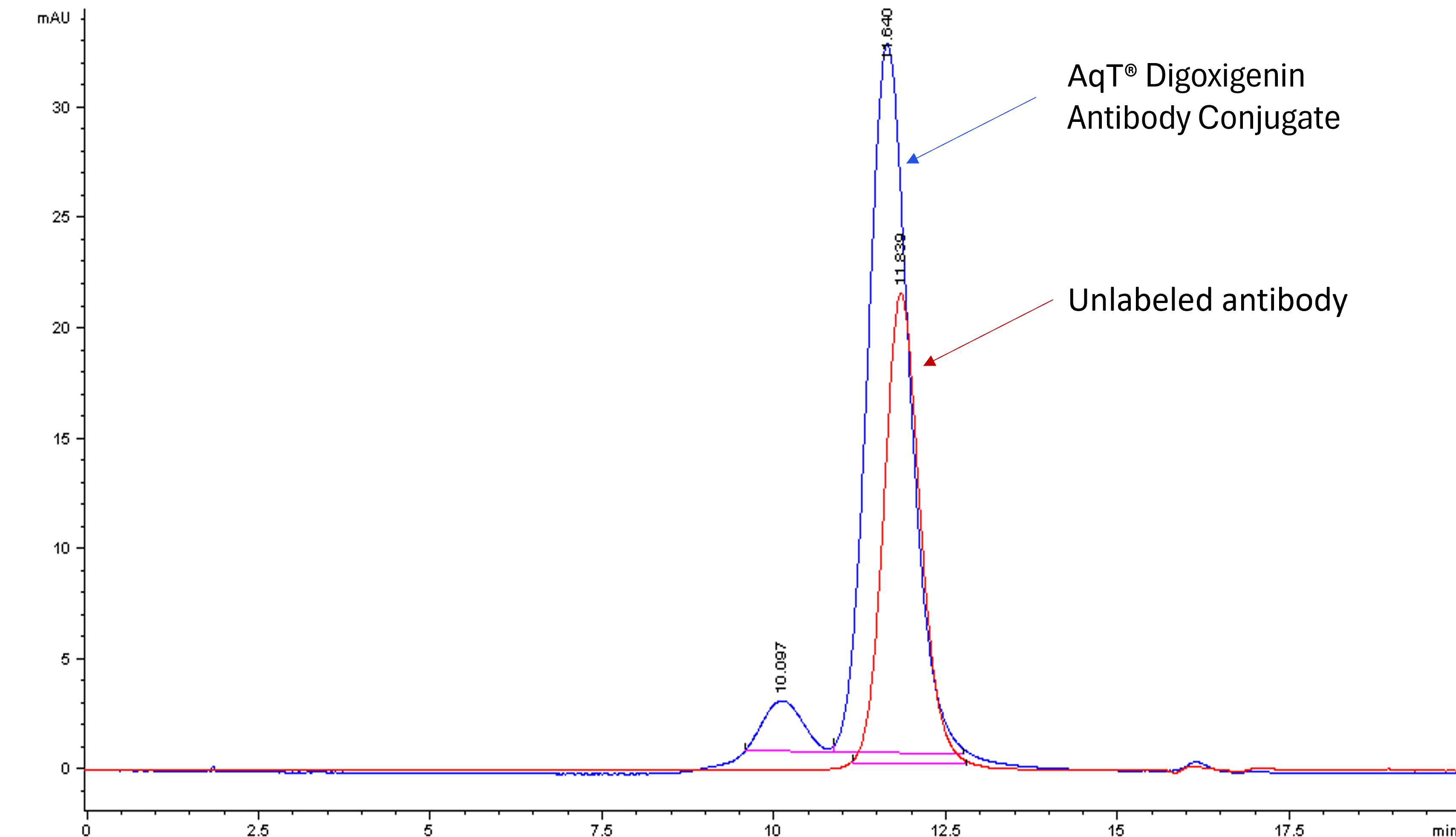
The figures below show an example of AqT® digoxigenin labeling of a human IgG1 monoclonal antibody. Using AqT® engineering, a high average degree of labeling (DOL: 6.9) was achieved despite the extreme hydrophobicity of the digoxigenin core. The extent of labeling was assessed by HIC HPLC (Figure 1). Apparent molecular weight and aggregation were evaluated by size-exclusion chromatography (SEC)-HPLC (Figure 2).
AqT® digoxigenin labeling with an average DOL of 6.9 shows a slight shift in apparent MW, and the conjugate has a hydrodynamic volume slightly smaller than the native antibody. Only 10% aggregation is observed after labeling at this such high loading. This confirms that AqT® labeling largely maintains antibody properties.
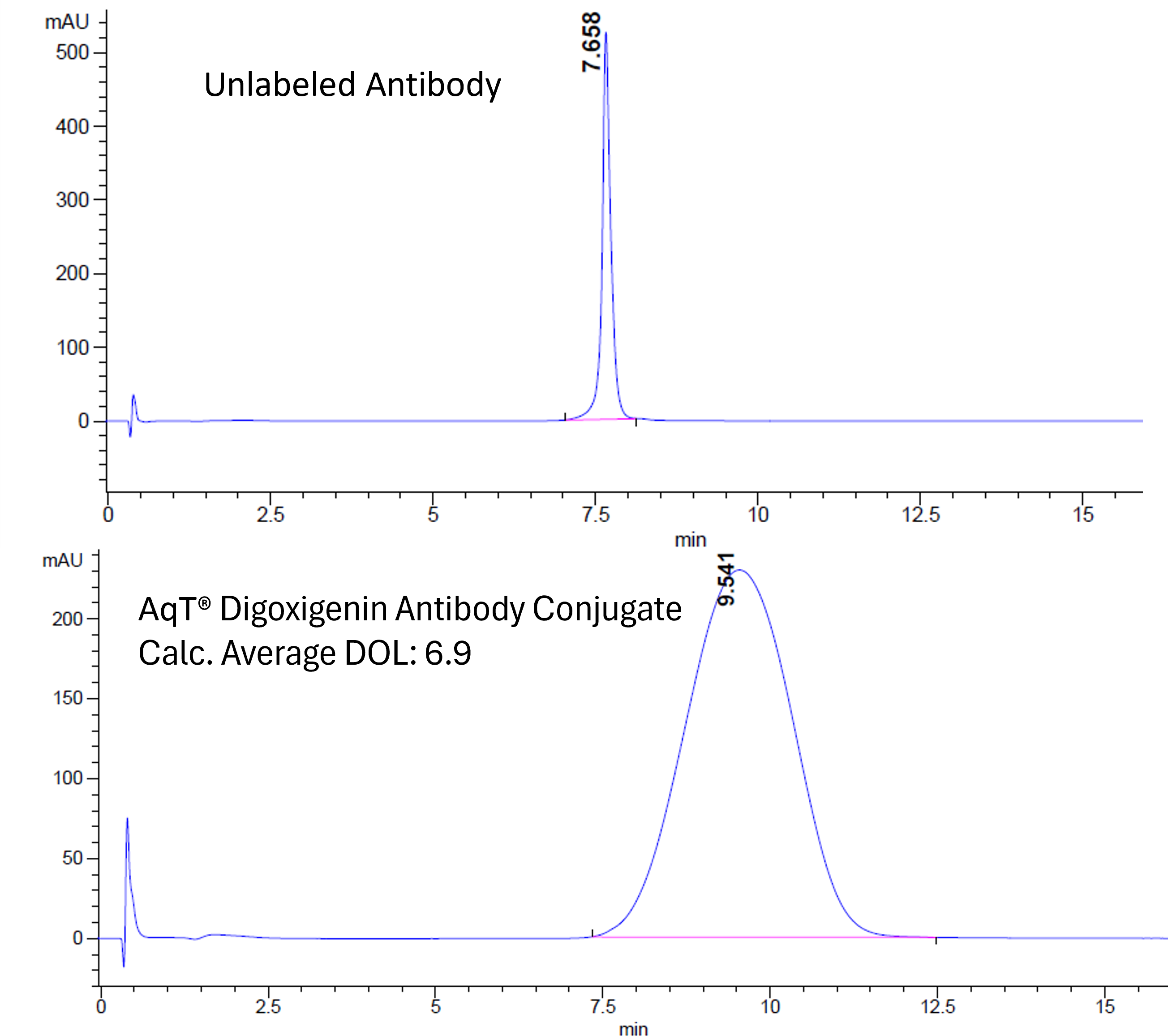
Figure 2: Overlay of SEC HPLC analysis of antibody and AqT® digoxigenin antibody conjugate.
Other Details
The information in this document is subject to change without notice. CellMosaic® assumes no responsibility for any errors or omissions that may appear in this document. Under no circumstances shall CellMosaic be liable, whether in contract, tort, warranty, or under any statute or on other legal theory, for special, incidental, indirect, punitive, multiple, or consequential damages related to or arising from this document, including but not limited to the use of this product thereof.CellMosaic’s products, including but not limited to AqT®, NeIon™, oxLink™ and sxLink™ (collectively, the “Products”), are covered by, or are the subject of, one or more issued patents and pending patent applications, including without limitation: 9,688,663 B2; 8,907,079 B2; 9,511,150 B2; 9,907,854 B2; 9,346,833 B2; 9,518,067 B2; CN104066451B; CN107043339B; CA 2,841,313; EP 2734238 B1; AU2012284055; JP 6240599 B2; and corresponding foreign patents and patent applications. The purchase of the Product conveys solely a limited, non-exclusive, non-transferable, non-sublicensable license to use the Product only for the purchaser’s internal research and development purposes. No right or license is granted, either expressly or by implication, estoppel, or otherwise, under any patent, trademark, copyright, or other intellectual property right of CellMosaic, except as expressly stated herein. Any commercial use of the Product is strictly prohibited without a separate written agreement from CellMosaic. Prohibited commercial uses include, without limitation: (a) the sale, lease, licensing, distribution, or other transfer of the Product or any materials derived from or produced using the Product; (b) the sale, lease, licensing, or other grant of rights to use the Product or any materials derived from or produced using the Product; and (c) the use of the Product to perform services for a fee or other consideration for third parties, including without limitation contract research, screening services, or diagnostic or therapeutic applications. For information regarding commercial licensing or collaboration, please contact us at info@cellmosaic.com.