Product Description
| Download Documents | |
| SDS | USER MANUAL will be supplied to customer |
AqT® Biotin Protein Labeling kit is a proprietary biotinylation kit developed at CellMosaic® for obtaining high quality and high loading of biotinylated protein with minimal or no aggregation.
AqueaTether® (AqT®) linkers are proprietary biomaterials developed at CellMosaic for labeling and conjugating biomolecules with highly hydrophobic small molecules, improving bioconjugate performance in downstream applications. For example, biotin is poorly water-soluble and prone to aggregation; incorporation of the AqT® linker enhances its solubility, enabling labeling with minimal or no organic solvent. The linker’s hydroxyl groups form hydrogen-bond networks with surrounding water, creating a stabilizing microenvironment that reduces biotin–biotin interactions and prevents aggregation or precipitation over time. This allows proteins to be labeled with high levels of AqT®-biotin without altering their properties, while potentially improving stability against enzymatic degradation and preserving activity. The AqT® Biotin Protein Labeling Kit (Surface Amines) leverages this technology to produce high-quality, low-aggregation conjugates using a super-hydrophilic, water-soluble, charge-neutral AqT® linker that also minimizes non-specific interactions with proteins and surfaces.
For a detailed description of the AqT® technologies, please see our technology section.
The kit is designed to label any protein with AqT® biotin via surface amines on the protein. The user supplies the protein. The kit includes AqT® Biotin Acid, which is activated within 15 minutes and then coupled directly to the protein in one step. The labeled product is subsequently purified to remove any unreacted AqT® Biotin Acid.
This kit provides materials to label 6.67 to 20 nano-mole (nmol) of one (CM86143x1) or three (CM86143x3) protein samples (≥20 KDa) with AqT® Biotin Acid. Customer can request a recommendation for the conjugation if the protein has a special feature, a less than 6.67 nmol of protein, or less than 20KDa MW of protein or peptide to be labeled. CellMosaic provides additional accessory tools, such as buffers, standards, and reagents, for protein research. We also offer fee-based support services for customers who require assistance with final conjugate analysis by HPLC and determination of the DOL.
Requirement for Protein
- MW: ≥ 20 KDa
- Preferably >90% pure by gel electrophoresis
- Total amount: 6.67-20 nmol protein content as measured by UV.
Key Features
- Advanced AqT® linker technology: Utilizes CellMosaic’s proprietary, super-hydrophilic, water-soluble, charge-neutral AqT® linker to minimize fluorophore stacking and protein aggregation.
- Tunable biotin loading with retained protein properties: Tunable loading from low to high, depending on your need, with no or minimal aggregation.
- All-inclusive kit with validated analytical method: Provides sterilized buffers and tubes, and filters for digoxigenin labeling. Also includes method needed for concentration measurement and loading determination.
- Fast, user-friendly workflow plus optional support: One-step labeling and purification in ~2 hours (≤1 hour hands-on or 0.5 h without purification), with optional post-conjugation services at CellMosaic® for analytical characterization and DOL determination.
Applications and Advantages of Highly Loaded AqT® Biotinylated Proteins
- Immunoassays (ELISA): Used as detection or capture reagents in ELISA formats; high biotin loading enhances signal amplification via streptavidin conjugates, while hydrophilicity reduces nonspecific binding and background.
- Flow Cytometry: Enables flexible labeling with streptavidin fluorophores; high biotin density increases signal intensity per binding event, and hydrophilicity minimizes aggregation while preserving protein activity.
- Immunohistochemistry (IHC) / Immunocytochemistry (ICC): Compatible with avidin–biotin complex (ABC) systems; high labeling improves detection sensitivity for low-abundance targets, while hydrophilicity enhances tissue penetration and staining uniformity.
- Western Blotting: Provides sensitive detection with streptavidin-based reporters; high biotinylation improves signal strength and detection limits without compromising specificity.
- Immunoprecipitation (IP) / Pull-Down Assays: High biotin loading improves capture efficiency on streptavidin-coated beads; multivalent binding ensures stable interactions, while hydrophilicity reduces nonspecific binding.
- Biosensor and Surface Immobilization (e.g., SPR, BLI): Enables strong and stable immobilization on streptavidin surfaces; high biotin density increases surface loading and sensitivity, while hydrophilicity improves accessibility and reproducibility.
- Magnetic Bead–Based Assays and Cell Isolation: Supports efficient capture using streptavidin beads; high labeling enhances binding capacity and recovery, while reduced aggregation improves sample integrity and performance.
- Microarrays and Multiplex Assays: Facilitates dense and stable protein immobilization; high biotin loading increases assay sensitivity and dynamic range, while hydrophilicity minimizes spot variability and background noise.
Labeling Chemistry
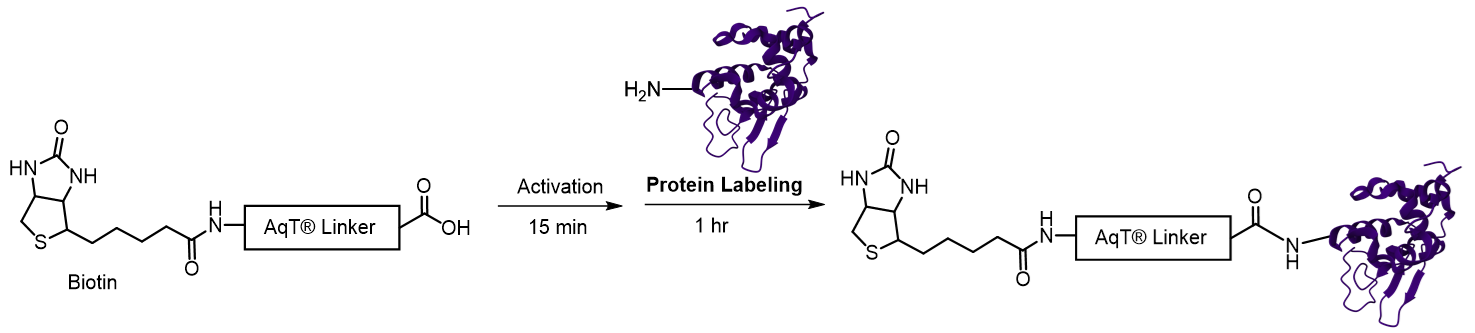
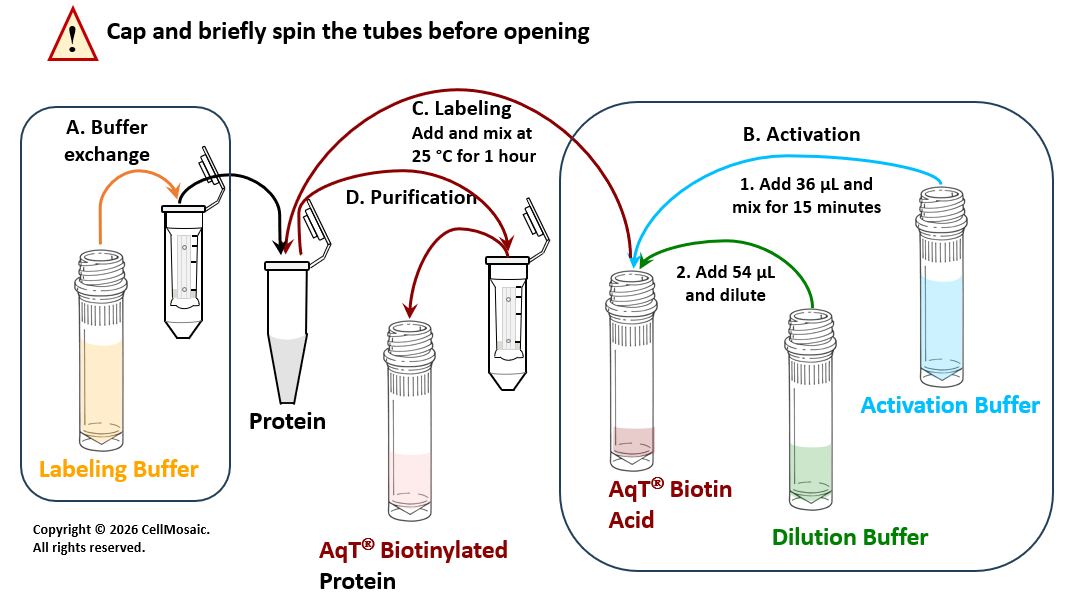
Protocol
Schematic workflow diagram for preparing AqT® biotin labeled protein within 2 hours, starting with 20 nmol of protein. (Reagent volume will vary if the amount of protein is less than 20 nmol).
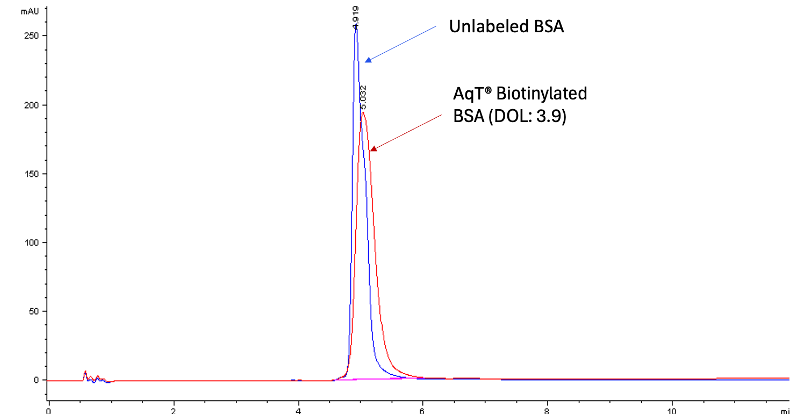
Characterization of AqT® Biotinylated Protein by Reversed Phase HPLC
For biopolymers labeled with very hydrophobic small molecules such as biotin, reversed phase HPLC can also be used to assess the extent of the biotinylation.
Figure 1 shows an example of AqT® Biotin labeling of BSA. Using AqT® engineering, an average DOL (3.9) was achieved. The hydrophilicity of the AqT® linker largely maintains the retention time of the conjugate peak close to the native BSA, with minimal peak broadening (indicating greater homogeneity).
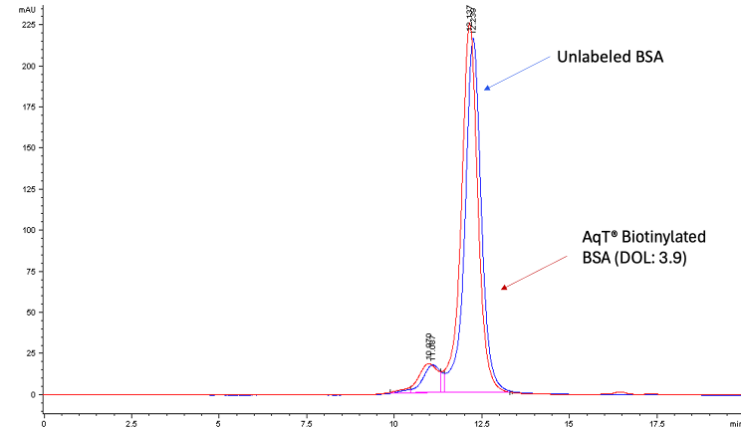
Characterization of AqT® Biotinylated Protein by SEC HPLC
SEC separates the conjugates by apparent MW or size in aqueous solution. In general, higher-MW species elute earlier. SEC is also a useful tool for assessing the level of aggregation in a biopolymer. By comparing the SEC profiles of an unlabeled protein and a biotin labeled protein, you can estimate the extent of aggregation in the labeled protein.
Figure 2 shows an example of AqT® biotinylated BSA. AqT® biotin labeling with an average DOL of 3.9 shows a slight shift in apparent MW, and the conjugate has a hydrodynamic volume slightly higher than the native protein. No additional aggregation is observed after labeling. This confirms that AqT® labeling largely maintains protein properties.
Frequently Asked Questions:
If you can’t find the answer you’re looking for or need information on general topics, please visit the main Frequently Asked Questions (FAQs) section.
Other Details
The information in this document is subject to change without notice. CellMosaic® assumes no responsibility for any errors or omissions that may appear in this document. Under no circumstances shall CellMosaic be liable, whether in contract, tort, warranty, or under any statute or on other legal theory, for special, incidental, indirect, punitive, multiple, or consequential damages related to or arising from this document, including but not limited to the use of this product thereof.CellMosaic’s products, including but not limited to AqT®, NeIon™, oxLink™ and sxLink™ (collectively, the “Products”), are covered by, or are the subject of, one or more issued patents and pending patent applications, including without limitation: 9,688,663 B2; 8,907,079 B2; 9,511,150 B2; 9,907,854 B2; 9,346,833 B2; 9,518,067 B2; CN104066451B; CN107043339B; CA 2,841,313; EP 2734238 B1; AU2012284055; JP 6240599 B2; and corresponding foreign patents and patent applications. The purchase of the Product conveys solely a limited, non-exclusive, non-transferable, non-sublicensable license to use the Product only for the purchaser’s internal research and development purposes. No right or license is granted, either expressly or by implication, estoppel, or otherwise, under any patent, trademark, copyright, or other intellectual property right of CellMosaic, except as expressly stated herein. Any commercial use of the Product is strictly prohibited without a separate written agreement from CellMosaic. Prohibited commercial uses include, without limitation: (a) the sale, lease, licensing, distribution, or other transfer of the Product or any materials derived from or produced using the Product; (b) the sale, lease, licensing, or other grant of rights to use the Product or any materials derived from or produced using the Product; and (c) the use of the Product to perform services for a fee or other consideration for third parties, including without limitation contract research, screening services, or diagnostic or therapeutic applications. For information regarding commercial licensing or collaboration, please contact us at info@cellmosaic.com.